The changes affect the structure and functions of microbial cells. 
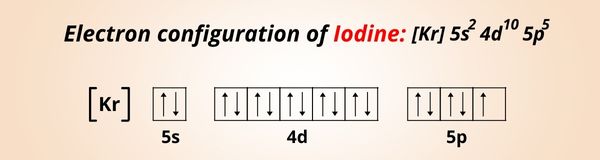
Iodine binds with proteins to cause their denaturation via several mechanisms, e.g., by oxidizing the SH groups in cysteine and methionine residues and preventing hydrogen bonding between the amino groups of arginines and histidines and the phenol groups of tyrosines. While antibiotics are localized in a particular site, iodine simultaneously affects all structures of a microbial cell. Iodine possesses antimicrobial and antiseptic properties. Complexation with polymeric carriers increases the solubility of molecular iodine, facilitates its prolonged release, and decreases the steady-state concentration of free iodine. Natural and synthetic water-soluble polymers and nonionic surfactants are broadly used as solubilizing agents. Iodophors are chemical complexes that contain a mixture of molecular iodine, iodide ions, and a solubilizing agent. In the early 1950s, a “conquest” of iodine started with investigating its complexation with certain polymers to yield compounds of a new class, which are known as iodophors. However, its use was limited by several undesirable factors, such as irritation, an increase in sensitivity, staining of biological and artificial surfaces, a low solubility in water, and a high vapor pressure. Iodine has long been used as an antiseptic to prevent and treat a broad range of infections. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
